Weekly Tech+Bio Highlights #4
ALSO: The Most Comprehensive Dataset of Human Proteome; AI-designed Drug Candidate is Patented in Japan, The Largest Foundation Model to Transform RNA Research: An Interview with Prof. Brendan Frey;
Hi! I am Andrii Buvailo, and this is my weekly newsletter, ‘Where Tech Meets Bio,’ where I talk about technologies, breakthroughs, and great companies moving the biopharma industry forward.
If you've received it, then you either subscribed or someone forwarded it to you. If the latter is the case, subscribe by pressing this button:
Now, let’s get to this week’s topics!
News Highlights
💰 J.P. Morgan Life Sciences Private Capital closes its inaugural biotechnology venture capital fund with over $500 million in commitments, aiming to invest in innovative biotechnology companies across various stages and therapeutic areas.
📈 Tempus AI announces the pricing of its initial public offering (IPO), with 11,100,000 shares of Class A common stock at $37.00 per share, aiming to raise $410.7 million in gross proceeds, with trading set to begin on Nasdaq under the ticker "TEM" on June 14, 2024.
Tempus AI is a technology company advancing precision medicine through the practical application of artificial intelligence in healthcare.
🏛 In global first, Cuorips, an Osaka University-affiliated startup, will seek approval from Japan's Government for its iPS cell-derived cardiac tissue sheet, a less invasive treatment for heart disease, after successful clinical trials.
🤝 Hoth Therapeutics partners with Wise Systems International to leverage Nvidia's BioNeMo AI platform for generative AI applications in drug discovery, enhancing their pipeline from target identification to lead optimization.
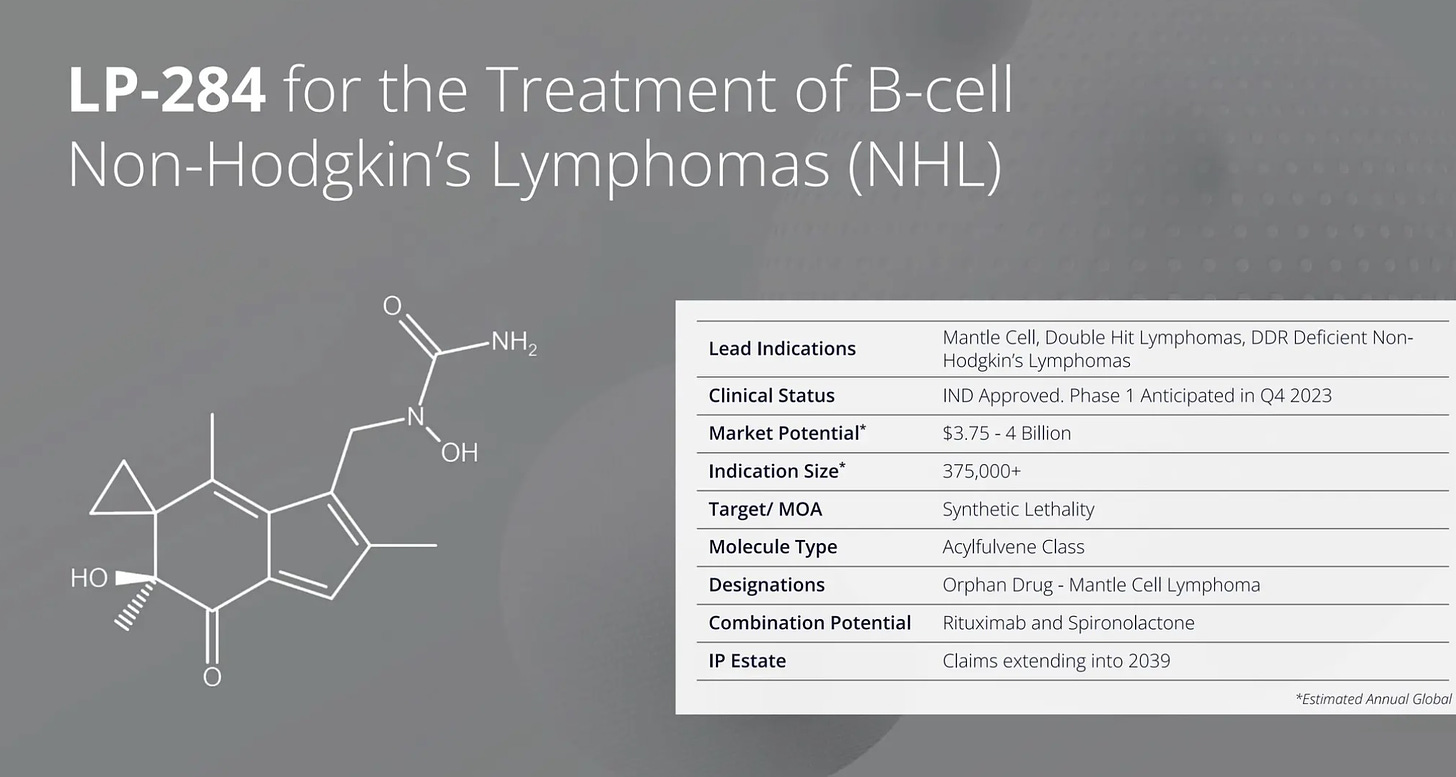
🔬 Lantern Pharma's AI-designed oncology drug candidate LP-284, currently in Phase 1 trials for non-Hodgkin’s lymphoma and certain sarcomas, receives a composition of matter patent in Japan, following previous patents in two other countries.
💰 Foresite Capital raises $900M for its sixth fund to invest in healthcare and life science startups at all stages, focusing on precision therapeutics, life science infrastructure, and healthcare delivery.
🔬 FluoSphera and Revvity announce a collaboration to develop a multiplexed selectivity assay for in vitro drug discovery, combining FluoSphera’s 3D cell culture system with Revvity’s HTRF assays and PhenoVue imaging reagents to evaluate drug target selectivity across multiple tissue types.
📈 QuantumPharm, operating as XtalPi Inc., a drug research company using AI and robotic automation, commenced trading on the Hong Kong Stock Exchange, raising approximately $126.8 million, the third-largest IPO in Hong Kong this year.
🚀 Santa Ana Bio, Inc. launches with $168M in Series A and B funding to develop precision therapies for autoimmune and inflammatory diseases, leveraging advanced proteomic, transcriptomic, and genomic techniques for targeted treatments.
🏛 Ultragenyx plans to file for accelerated approval of UX111, an AAV gene therapy for Sanfilippo syndrome Type A (MPS IIIA), after reaching an agreement with the FDA that cerebral spinal fluid heparan sulfate can be used as a surrogate endpoint.
I am glad to announce our media partnership with HLTH Europe!
As Europe’s premier health event, HLTH Europe 2024 will unite every segment of the continent’s health ecosystem in Amsterdam, on June 17-20.
The inaugural 2024 event will feature:
• 3,500+ attendees
• 400+ speakers
• 300+ sponsors
• Representation from 50+ countries
Prominent media outlets like BBC News, DER SPIEGEL, and Sifted will be in attendance, alongside global sponsors including GE HealthCare, Google Health, Microsoft, Philips, Novo Nordisk, and Bayer.
Join us to discuss critical topics such as health reimbursement, preventative care measures, chronic disease management, and regulatory changes.
Introducing the Most Comprehensive Dataset of Human Protein Structures
Understanding the molecular landscape of human proteins is crucial for advancing biomedical research, particularly in drug discovery and protein function prediction.
A recent publication in Scientific Data by Michael Hetmann and colleagues introduces a groundbreaking dataset of predicted protein structures for 42,042 distinct human proteins, including splicing variants, derived from the UniProt reference proteome UP000005640.
This dataset leverages state-of-the-art AI-driven modeling tools—AlphaFold 2, OpenFold, ESMFold, and homology modeling via Innophore’s CavitomiX platform—integrated within NVIDIA’s BioNeMo platform.
The result is a highly comprehensive and diverse collection of human protein structures, available in both native and refined formats to support a wide array of research applications.
Key Takeaways
The dataset includes 42,042 protein structures, covering 99.72% of the targeted human protein sequences, significantly extending the existing structural information provided by previous efforts such as the AlphaFold database.
By employing multiple AI-guided structure prediction tools—AlphaFold 2, OpenFold, and ESMFold—alongside homology modeling from CavitomiX, the dataset ensures high structural diversity, which is critical for various applications like virtual docking and molecular-dynamics simulations.
The dataset is available in both native and refined formats. The refined version includes structures with low-confidence regions removed and energy minimization applied, enhancing the utility for specific applications such as ligand binding site analysis.
Ligands were integrated into AI-generated models based on structural similarity to homology models, providing valuable information about potential binding sites. This feature is particularly important for drug discovery efforts, as it helps identify interactions between small molecules and proteins.
The dataset includes detailed quality control measures, such as per-residue pLDDT scores for AI-predicted models and z-scores for homology models. Comprehensive statistics and model-specific information are provided to help researchers critically assess the reliability of each structure.
Oncology drug candidate, designed with help of AI, gets patended in Japan
News comes from Dallas-based clinical stage biotech Lantern Pharma Inc. (Nasdaq: LTRN), a developer of AI platform for drug discovery and repurposing RADR®.
Key takeaways:
● LP-284 is currently in a Phase 1 clinical trial, having been developed with guidance from Lantern’s AI platform, RADR®, as a potential therapy for relapsed or refractory non-Hodgkin’s lymphoma and certain genomically defined sarcomas.
● Lantern estimates that LP-284 can have the potential to improve outcomes for 40,000 to 80,000 patients with blood cancers annually, with a global annual market potential of $4 Billion USD.
● LP-284 is the third molecule brought to clinical trials by Lantern Pharma using insights and support from RADR®.
● Japan is the third country to date where a composition of matter patent has been issued for LP-284.
● The addition of the new patent further strengthens LP-284’s future clinical development and creates pathways for potential future geographic partnerships.
Pioneering the Largest Foundation Model to Transform RNA Research: An Interview with Brendan Frey
Toronto-based “techbio” company Deep Genomics has just annouced major milestones with the opening of a new office and lab facility in Cambridge, Massachusetts, and the expansion of its Toronto office.
In this interview, we sit down with Prof Brendan Frey, Founder and CIO of Deep Genomics, to delve into his vision for the future of RNA therapeutics and the integral role of AI in advancing biological research.
Frey discusses how the recent expansions and new executive hires will enhance Deep Genomics' capabilities in decoding RNA biology and designing innovative therapeutics. He also shares insights into how their AI platform, BigRNA, is set to transform genomic R&D, reduce risks, and accelerate the development of new treatments.
Andrii: With the increasing role of AI in biotech, how do you see Deep Genomics positioning itself within the industry? What sets your approach apart from other companies working on AI-driven drug discovery and development?
Brendan: Productivity in R&D for the Life Sciences industry continues to be its greatest challenge. By pioneering a novel new approach to early stage R&D, we’re charting a unique path as the leaders of a new category of TechBios, and guiding improved genomic R&D as a result.
Whereas the tech world has a rich open source history, this ethos is relatively new to the pharmaceutical industry. Organizations that are built from the ground up with technology as the driving force enable seamless integration and collaboration between biologists, chemists, and AI researchers.
We are placing AI at the forefront of genomic R&D. Since 2018, we’ve coined terms at Deep Genomics such as ‘programmable medicines’, ‘digital medicines’ and ‘AI-biology multilingual teams’, which are now commonly used terms. Tech is not the new factor here – but by moving forward with our TechBio approach, we can more readily acknowledge AI’s vital role in the pharma industry.
Andrii: RNA biology is a complex and rapidly evolving field. What initially drew you to focus on RNA, and why do you believe it holds such significant potential for therapeutic development?
Brendan: We often say RNA is essentially the ‘software of biology’, making it the ideal place to intervene with AI. With RNA's optimal fundamental design, the availability of the data available, and big steps forward made in computing power – RNA holds massive potential for transforming not just the drugs in the market – but the way we think about pharma and R&D.
Drugs aligned with proteins or biologics are neither well-matched to a computer, nor AI. RNA, however, is fundamentally digital - it’s made up of a sequence of letters. What could be better matched to a computer than a sequence of letters? Not only that, but unlike its DNA editing and gene therapy counterparts, RNA therapy enables tunability and reversibility, offering unique advantages in specific therapeutic settings.
Lastly, we have more data for RNA than any other kind of therapeutic modality. This positions RNA as the key therapeutic route to pave the way for transformative developments in the pharma industry. With hundreds of petabytes of information, and a field that is so complex, the key next step in the RNA journey was developing AI that can untangle these complex relationships, accurately prioritize therapeutic opportunities, and work at scale across the entire genome.
That’s what Deep Genomics’ AI foundation model, BigRNA, aims to do. With the power of AI growing exponentially, we can use this technology to bridge the fundamental gap between our ability to measure mutations, understand their consequences on molecular and cell biology, identify targets, and create precision treatments. Our AI foundation model is paving the way to decode RNA biology and program life changing medicines.
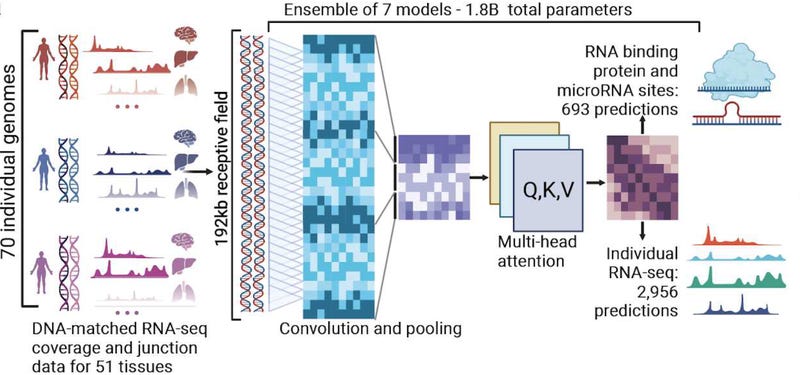
Andrii: BigRNA is a groundbreaking AI foundation model with 1.8 billion tunable parameters built from 1 trillion genomic signals. Could you walk us through some of the most exciting technological breakthroughs that have made this model possible, and what unique capabilities it brings to RNA research?
Brendan: As the world’s first foundation model for RNA biology, BigRNA is changing the game for how we approach R&D.
It takes as input any gene sequence and can accurately predict thousands of different molecular biology outcomes, enabling the discovery of targets, disease mechanisms and RNA therapeutics across a wide range of diseases, tissue types, and genetic architectures, including N=1, ultra rare, rare and common diseases.
However, we didn't get to BigRNA overnight. Prior to BigRNA, we invested intensively in developing 40 different machine learning systems, one for each biological process. But now, our BigRNA foundation model enables us to train on everything, everywhere, all at once. Different tasks benefit from one another, achieving synergies where predictions for different mechanisms and molecules are coordinated.
BigRNA enables rapid exploration of RNA biology at scale, from species, tissues, cell models, variants, genes, oligos, and editing, including mechanisms of splicing, polyadenylation, protein/microRNA binding, in vitro validation, and support for multiple therapeutic modalities. Overall, BigRNA has shown great potential in accelerating drug discovery.
The past year has been groundbreaking. Introducing BigRNA has ushered in a major transformation for us, one that has and will continue to pave the way for immense growth in the pharma industry.
When we plot the capability of AI systems across industries, we see exponential growth. We’re proud to be pioneering this next phase of tech in pharma, and are already looking on the horizon for more ways we can further accelerate drug discovery transformation as a major TechBio player.
Andrii: Deep Genomics aims to use AI to decode RNA biology and design therapeutics. What are some of the most promising therapeutic areas or diseases where you see BigRNA making the most significant impact in the near future?
Brendan: BigRNA is groundbreaking because it enables the discovery of targets across a wide range of diseases – from common diseases, to rare and ultra rare. Our AI foundation model captures an already wide range and allows for support for multiple therapeutic modalities.
For example, in Wilson’s Disease, BigRNA evaluated a patient mutation that changes an amino acid in ATP7B, a copper-binding protein that is absent in Wilson patients. It predicted that this change will not affect how the protein works, but instead that it alters the regulatory genome, disrupting an instruction in the genome that tells cells how to make that protein, causing it to not be made at all. This is just one example among many.
With this in mind, we’re expecting BigRNA to continue to make an impact on a wide range of diseases in the future, and are most excited to see how our technology finds more ways to solve these issues in a more effective and efficient way.
Andrii: Developing and deploying such advanced AI models in biotech must come with its set of challenges. What are some of the major hurdles Deep Genomics has faced, and how have you and your team overcome them to achieve your current success?
Brendan: With BigRNA, we’ve taken the field a leap forward with rapid exploration of RNA biology at scale, in vitro validation, and support for multiple therapeutic modalities.
We’re working on improving our resolution, to go from 128bp to 1bp, which will enable the discovery of new biology, such as the effects of indels. We’re improving our accuracy to enhance our prediction performance, and moving our validation data into DNA/RNA editing and mRNA.
Another vital piece here relates to how our technology can best support the community. We’re constantly asking ourselves: How can we help with diagnostics? Making these discoveries is mind-blowing and opens doors to groundbreaking progress in RNA therapy – but it's when we see the positive effect on the community in real life use cases that we’ll know we’ve done something truly great for pharma.
Andrii: The biotech industry is rapidly evolving with advancements in AI and genomic research. In your opinion, what are the most exciting trends or innovations we can expect to see in the next decade, and how do you see Deep Genomics contributing to these advancements?
Brendan: One of the more exciting things we’re looking to focus on in the next decade, aside from the results of foundation models like BigRNA, is a change to R&D culture - one that features tighter partnerships between computational scientists and biological scientists, and between pharmas and a new class of TechBios.
In the past, both pharmaceutical companies attempting to leverage AI and AI companies attempting to apply their models to drug design have faced significant challenges. These setbacks are rooted in a failure to bridge the cultural and language barriers between the disparate disciplines involved.
Adopting AI foundation models requires a fundamental mindset shift that fosters seamless collaboration across domains. At the core of this transformation is the need for "multilingualism" – the ability to facilitate effective dialogue between experimental biologists and AI researchers. Multilingualism has to be a core value, recognising that true breakthroughs in AI-driven drug discovery can only be achieved when researchers transcend the silos of their respective fields.
Merely bolting on AI capabilities to existing pharmaceutical processes is not sufficient; AI must be woven into the very fabric of the drug discovery and development process. Generating the wide and general outputs that AI foundation models promise requires a level of deep integration and true collaboration that cannot be achieved through traditional approaches.
This new approach necessitates a unique kind of partnership – one that brings together the domain expertise of pharmaceutical companies with cutting-edge machine learning and the AI capabilities of technology-driven organizations.
Successful partnerships will accelerate the discovery of life-saving therapies.
Andrii: As someone who has made significant contributions to both AI and genomic medicine, what personal experiences or insights have most shaped your leadership at Deep Genomics? How do you stay motivated and inspired to push the boundaries of what's possible in this field?
Brendan: Studies show that 95% of rare diseases do not have approved treatment. I faced the hardships of this statistic in my family 20 years ago, and it’s what kick started my journey. I wanted to find a more efficient and effective way of helping people who are in the situation I was in, and helping the world have access to better medicines.
While others have struggled to connect AI approaches and biology-led science, Deep Genomics has purpose-built the team, culture and tools to combine AI and biology science for groundbreaking results.
We take a hybrid leadership approach, fusing the diverse skills, backgrounds, and experiences of our team, along with the support of the Scientific Advisory Board – to drive our innovations forward. We’ve developed a versatility that proactively addresses the issues facing pharma, placing multilingualism as a core value at the center of our discoveries. With that focus, we’ve crossed the barriers between computational and experimental biologists. In an industry that can thrive best by sharing information and collaborating, breaking down silos not only pushes the boundaries of what’s possible, but is what makes me confident about what more we’ll continue to create and discover.
I continue to be impressed with the discoveries of team members at Deep Genomics, as they stretch frontiers. I stay motivated knowing that parents, family, and friends will be able to avoid what I experienced two decades ago, with technologies like BigRNA. Knowing that we’re fundamentally changing the way pharma operates by integrating AI into RNA is all the inspiration I need, as we continue to pioneer more innovative ways to help the people, families, and communities that need it.
Some Topical Newsletters You May Like:
AI Foundation Models in Biotech: New Paradigm
How Industry Embraces Organ-on-Chips: A 2024 Status Report
A Landscape of Novel Antibody-Drug Conjugates
11 Biopharma Trends to Watch in 2024
Using Quantum-Enhanced AI to Design Cancer Drugs For the First Time
It’s Been a Decade of AI in the Drug Discovery Race. What’s Next?