Weekly Tech+Bio Highlights #5
ALSO: Google Unveils Tx-LLM for Drug Discovery; Quantum Computing for Clinical Trial Optimization; Anonymizing 1.8 Trillion Records for Biomedical Research; Gen AI is all the Rage in Healthcare
Hi! I am Andrii Buvailo, and this is my weekly newsletter, ‘Where Tech Meets Bio,’ where I talk about technologies, breakthroughs, and great companies moving the biopharma industry forward.
If you've received it, then you either subscribed or someone forwarded it to you. If the latter is the case, subscribe by pressing this button:
Now, let’s get to this week’s topics!
News Highlights
✔️ Roche gains FDA 501(k) clearance for its Digital Pathology Dx system, which aids pathologists in reviewing and interpreting digital images of pathology slides for patient diagnosis, featuring the VENTANA DP 200 slide scanner and digital pathology workflow software.
Roche also partnered with Ascidian Therapeutics on RNA exon editing drugs, investing $42 million upfront with up to $1.8 billion in milestone payments.
💰 Enveda Biosciences secures $55M in a Series B2 round, raising its total capital to $230M, with participation from new and existing investors including Premji Invest, Microsoft, and Kinnevik.
🔍 Iambic Therapeutics' AI platform, NeuralPLexer, showcased in Nature Machine Intelligence, surpasses systems like AlphaFold2 in predicting protein-ligand complex structures. Their ProPANE platform utilizes a graph neural network for optimizing multiple drug properties.
On June 18, Iambic Therapeutics annouced an additional $50M round led by Mubadala Capital and Exor Ventures, for its lead program IAM1363 and upcoming IAM-C1, aimed at treating HER2-positive cancers and solid tumors respectively.
🔬 Insilico Medicine completes patient enrollment for its Phase IIa study in China on INS018_055 for idiopathic pulmonary fibrosis, with a robust dataset expected by late Q4 2024 and a Phase IIb proof-of-concept study planned for 2025.
☁️ Athos Therapeutics selects Vultr's private cloud and NVIDIA HGX H100 GPUs to enhance its AI drug discovery engine, integrating multi-omics data from over 25,000 patients to identify novel drug targets for autoimmune and chronic inflammatory diseases.
⚠️ FDA imposes partial hold on BioNTech/MedLink antibody-drug conjugate trial after three patient deaths, affecting new U.S. enrollments in Phase I trial for BNT326/YL202, an advanced cancer therapy. Current patients will continue treatment as the focus shifts to safer, lower doses.
🧠 Google Research and DeepMind introduce Tx-LLM, a drug discovery and therapeutic development tool fine-tuned from Med-PaLM 2, an AI designed to answer medical questions. Trained on 709 datasets for 66 tasks, Tx-LLM outperformed state-of-the-art models in 22 tasks and matched their performance in 21 others.
🔬 Insilico Medicine completes patient enrollment for its Phase IIa study in China on INS018_055 for idiopathic pulmonary fibrosis, with a robust dataset expected by late Q4 2024 and a Phase IIb proof-of-concept study planned for 2025.
🤝 Roche partners with Ascidian Therapeutics on RNA exon editing drugs, investing $42 million upfront with up to $1.8 billion in milestone payments.
🔬 Basecamp Research, in collaboration with the Ferruz Laboratory, launches ZymCTRL, the first open-source, text-based AI tool for enzyme design, enabling the creation of novel enzyme sequences for applications in therapeutics and sustainable industries.
🔬 GNQ Insilico, a portfolio company of Trenchant Technologies Capital, has successfully used its AI-driven platform to create digital twins of human patients and simulate the effects of an infertility drug, potentially enhancing drug discovery and reducing clinical trial costs and failure rates.
🔬 Yonalink completes its largest electronic health record (EHR)-to-electronic data capture (EDC) data transfer pilot across 21 medical centers in the US and Israel, streaming 93% of patient data with 100% accuracy, setting a new industry standard for clinical trials.
Google Unveils Tx-LLM for Drug Discovery
Google Research and DeepMind have introduced Tx-LLM, a new large language model (LLM) specifically designed to enhance drug discovery and therapeutic development. This model is an advanced version of Google's Med-PaLM 2, tailored to analyze a wide array of chemical and biological entities.
Why it matters: The Tx-LLM aims to significantly improve the efficiency of drug development by assisting in various stages, from efficacy and safety evaluation to target prediction and manufacturability.
Key Aspects: The model was trained using 709 datasets to perform 66 distinct tasks across the drug discovery spectrum. These tasks include:
Evaluating drug efficacy and safety.
Predicting molecular targets.
Assessing the ease of manufacturing drugs.
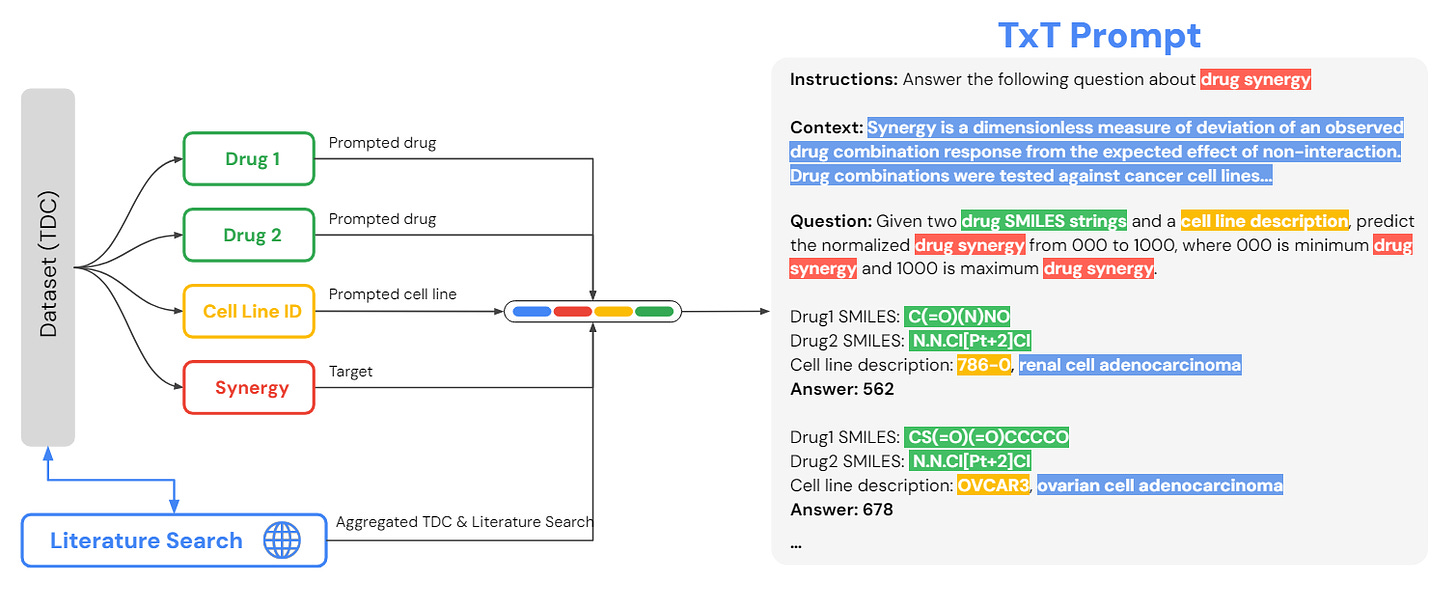
Tx-LLM utilizes the Therapeutics instruction Tuning (TxT) collection, which combines free-text instructions with representations of small molecules, such as SMILES strings.
SMILES (Simplified Molecular Input Line Entry System): A method that uses printable characters to represent molecules and chemical reactions.
TxT: Used to prompt and fine-tune Tx-LLM, enabling it to handle classification, regression, and generation tasks critical to drug discovery and development.
Performance and Innovation: Tx-LLM has demonstrated exceptional performance, achieving results that are at or near the state-of-the-art (SOTA) models for 43 out of the 66 tasks. It exceeded SOTA models in 22 tasks, showcasing its superior capability in:
Classification tasks.
Regression analysis.
Generative modeling.
Positive Transfer Learning: The model exhibited positive transfer between datasets with diverse drug types. For instance, training on datasets that include biological sequences has been shown to enhance the model's performance on molecular datasets. This indicates that Tx-LLM can effectively integrate and learn from a variety of data sources, improving its overall predictive accuracy.
What’s next: Tx-LLM holds promise as an end-to-end assistant for therapeutic development, allowing researchers to query a single model across multiple stages of the development pipeline. This integration could lead to faster and more efficient drug discovery processes.
Further Reading: For an in-depth understanding of Tx-LLM's architecture, training process, and potential applications, refer to the detailed paper published by Google Research and DeepMind.
Exploring Quantum Computing for Clinical Trial Optimization
Researchers from various leading institutions, including IBM Quantum, Deloitte Consulting, and the Cleveland Clinic, have explored the application of quantum computing to enhance clinical trial design and optimization and published findings in a pre-print titled "Towards quantum computing for clinical trial design and optimization: A perspective on new opportunities and challenges."
Why it matters: Clinical trials are critical for assessing the safety and efficacy of new drug candidates. However, high failure rates often stem from inadequate clinical model development and flawed protocol designs.
The design of clinical trials involves rigorous protocols to ensure patient safety and data quality. Key challenges include:
Patient Cohort Selection: Ensuring statistically significant event rates and tracking adverse events across diverse populations.
Trial Optimization: Balancing trial costs with achieving significant outcomes to support FDA approval.
Data Integration: Utilizing multi-modal clinical, demographic, and geographic data to assess patient fit individually.
Quantum Computing Solutions: Quantum computing, based on principles of quantum mechanics such as entanglement, superposition, and interference, holds potential to outperform classical computing in certain complex problems. The study focuses on three critical aspects where quantum algorithms can be applied:
Clinical Trial Simulations: Using quantum differential solvers to understand the mechanistic properties of drugs.
Site Selection: Employing portfolio-based quantum optimization to identify the best set of trial sites.
Cohort Identification: Utilizing quantum generative models to improve the selection process of patient cohorts.
Technical Details: The research leverages quantum optimization and quantum machine learning methods to enhance clinical trial design. Quantum algorithms can potentially address classically intractable problems, providing computational speed-ups in tasks such as:
Prime factorization.
Unstructured database searches.
Solving linear systems.
Computing topological invariants.
Quantum simulations.
Proposed Workflow: The study outlines a comprehensive framework using quantum computing for clinical trial optimization:
Protocol Design: Establishing trial parameters and using quantum solvers for enhanced understanding of drug properties.
Logistical Optimization: Informing site selection and cohort identification through quantum optimization methods.
What’s next: This pioneering work sets the stage for further research into quantum computational methods for clinical trial design. Future directions may include more extensive implementation of quantum algorithms and broader applications across different phases of clinical trials.
This Company Anonymized 1.8 Trillion Records to Support Biomedical Research
In the rapidly evolving life sciences and healthcare landscape, the secure and privacy-preserving exchange and linkage of health data is paramount. The topic of biomedical data collection and processing received a lot of attention during HLTH Europe 2024, where I was covering news for BiopharmaTrend, and one company that stood out to me in this field was Datavant.
Datavant is a developer of a technology that facilitates the secure, privacy-preserving exchange and linkage of health data.
Datavant was founded in 2017 and is headquartered in San Francisco, California. The company has raised $83 million in funding to date.

A 2022 survey by Savvy Cooperative, involving 1,000 patients and reported by the American Medical Association (AMA), highlighted the importance of data privacy. Over 92% of patients believe privacy is a fundamental right and that their health data should not be available for purchase.
Nearly 75% expressed concern about the protection of their personal health data, and only 20% felt they understood the full scope of who has access to their information. Responding to this mandate,
Datavant has developed solutions that aim to address concerns by patients but also make sure the relevant data is available for critical research purposes, ensuring privacy preservation while enabling secure health data exchange.
Data Tokenization is to the Rescue
At the heart of Datavant's technology is tokenization, a process that converts sensitive data, known as Personally Identifiable Information (PII), into a non-sensitive equivalent called a token.
This involves hashing and encrypting PII, transforming it into a token that cannot be reverse-engineered to reveal the original data. For example, a name like "John Smith" is always converted into the same unique token. Since different data sets may capture various PII elements, Datavant creates multiple tokens for a single piece of PII, ensuring comprehensive protection across diverse data environments. Advanced algorithms enable these tokens to be matched across different records without exposing the underlying PII, allowing for the secure linkage of patient records from disparate sources.
Datavant excels in data logistics, facilitating the seamless movement of health data from one point to another. Their technology integrates with over 500 data sources, including analytics companies, health systems, electronic health records (EHR) companies, and diagnostics labs. This integration breaks down data silos and facilitates comprehensive data exchange. Datavant’s solutions are embedded within the UK’s NHS GP system and the NHS Spine.
In North America, they work with over 75,000 clinics and hospitals, and their technology is deployed across more than a dozen countries, primarily in Europe and North America.
Ensuring privacy and compliance is a cornerstone of Datavant’s approach. Techniques such as anonymization, pseudonymization, and de-identification are used to protect patient identities. Datavant’s methods comply with key regulations, such as HIPAA in the United States, ensuring that all data exchanges meet stringent privacy and security standards.
Supporting Life Science and Healthcare Research with Anonimized Data
Record linkage is another critical aspect of Datavant’s technology. Using tokenization, Datavant can link records pertaining to the same individual across different data sets without revealing their identity. For example, data from a diagnostic lab and an EHR can be linked to create a comprehensive patient record. This capability is particularly beneficial in clinical trials, where linking real-world data with trial data provides valuable insights into patient outcomes without compromising privacy.
On the practical side of things, Datavant’s technology has significant implications for the life sciences sector. By supporting the linkage of real-world data with clinical trial data, Datavant enhances the ability to monitor patients over long periods without additional burden.
The company has supported over 100 clinical trials, enabling the compliant exchange of real-world data. Annually, they handle the anonymization and secure linkage of over a trillion patient records, ensuring privacy while facilitating valuable insights for life science and healthcare research.
Their solutions help in understanding real-world outcomes and costs of new treatments, supporting market access and reimbursement.
Datavant also facilitates digital engagement with patients, ensuring compliance while enabling the comprehensive use of generated data.
Healthcare Leaders Turn to Gen AI to Address Critical Gaps in Patient Care
A trove of insights came from the announcement during HLTH Europe 2024 of the Future Health Index (FHI) by Royal Philips (NYSE: PHG, AEX: PHIA).

This annual report, now in its 9th edition and currently the largest study of its kind, highlights the increasing reliance on virtual care and AI-enabled innovations to address significant challenges in the healthcare sector, such as workforce shortages, financial burdens, and growing demand for services.
Key highlights fron the FHI 2024 report:
AI and Automation: An overwhelming 92% of healthcare leaders believe that automation is crucial for addressing staff shortages by reducing the administrative burden on healthcare professionals. However, 65% of these leaders acknowledge skepticism among healthcare staff regarding the use of automation.
Virtual Care: Nearly 89% of healthcare leaders have observed a positive impact of virtual care in easing staff shortages, improving patient care delivery, and enhancing staff satisfaction.
Generative AI: The report indicates a significant interest in generative AI, with 85% of healthcare leaders currently investing in or planning to invest in these technologies to enhance clinical decision support and patient monitoring.
Staffing Challenges: Approximately 66% of healthcare leaders report increased burnout and mental health issues among their workforce, underscoring the need for digital solutions to improve efficiency and reduce workloads.
Financial Sustainability: Financial challenges are affecting patient care, with 81% of leaders noting an impact due to limited resources. Strategies to address these challenges include embracing subscription-based models for healthcare technology to reduce upfront costs.
Some Topical Newsletters You May Like:
AI Foundation Models in Biotech: New Paradigm
14 Foundation Models for Biology Research and Chemistry
How Industry Embraces Organ-on-Chips: A 2024 Status Report
A Landscape of Novel Antibody-Drug Conjugates
11 Biopharma Trends to Watch in 2024
Using Quantum-Enhanced AI to Design Cancer Drugs For the First Time
It’s Been a Decade of AI in the Drug Discovery Race. What’s Next?